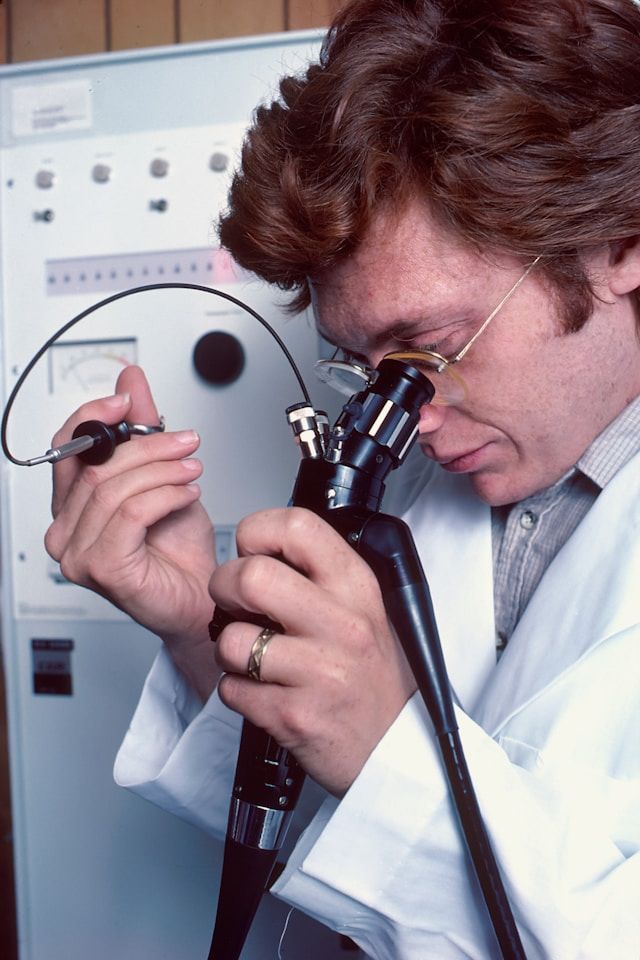
CMMS for Pharma Compliance: 5 Powerful Reasons to Boost Your GMP Readiness
GMP readiness depends on traceable maintenance and controlled changes. Learn 5 reasons CMMS improves pharma compliance, audits, and documentation.
Explore maintenance practices for pharmaceutical facilities focused on compliance, validation, and equipment reliability.

GMP readiness depends on traceable maintenance and controlled changes. Learn 5 reasons CMMS improves pharma compliance, audits, and documentation.

Cold room failures trigger deviations and audit findings. Learn 5 GMP-ready checks for sensors, alarms, defrost, door integrity, and documentation.

Audit readiness depends on disciplined maintenance records. Learn 7 GMP maintenance steps: PM control, traceability, change control, and review.
Freezer failures risk product, quality, and GMP compliance. Learn 5 proven ways pharma teams track freezer downtime and act before excursions.
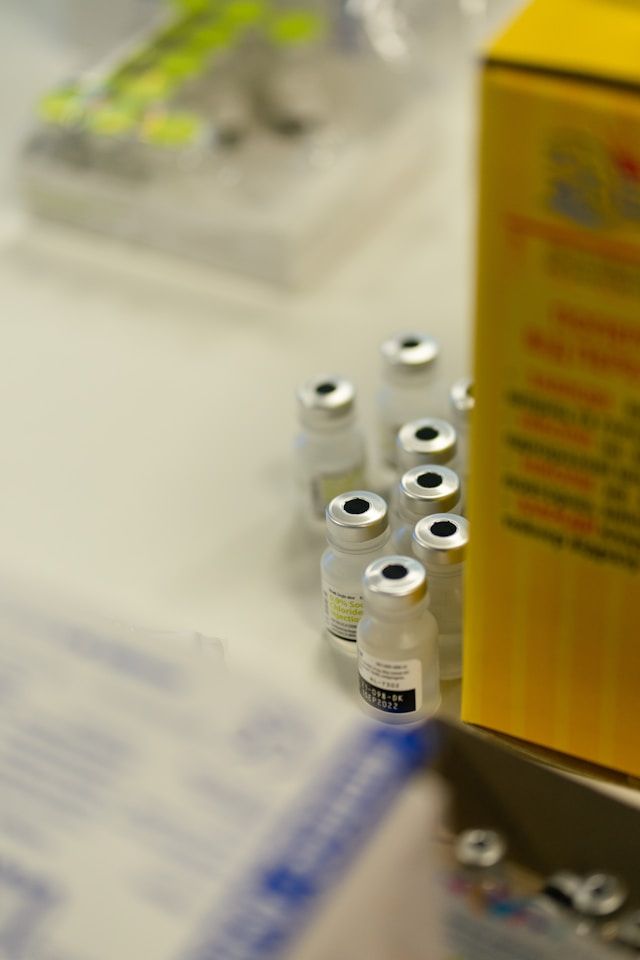
Vaccine cold chain reliability depends on maintenance controls. Learn 6 WHO PQS focus areas and the maintenance checks that keep storage compliant.